The following information is presented for educational purposes only. Medical Marijuana Inc. provides this information to provide an understanding of the potential applications of cannabinoids. Links to third party websites do not constitute an endorsement of these organizations by Medical Marijuana Inc. and none should be inferred.
At Medical Marijuana Inc., we are exploring new ways for marijuana and hemp to positively affect people’s lives, and one of the most important is medicine. With over 23,000 research publications in the past few decades dedicated to cannabinoids and the endocannabinoid system, there is overwhelming evidence that cannabis has the potential to impact the health of people around the globe. Medical Marijuana Inc. is actively developing and commercializing opportunities to integrate cannabinoids into mainstream medicines, in the face of the legal and regulatory barriers we face day to day.
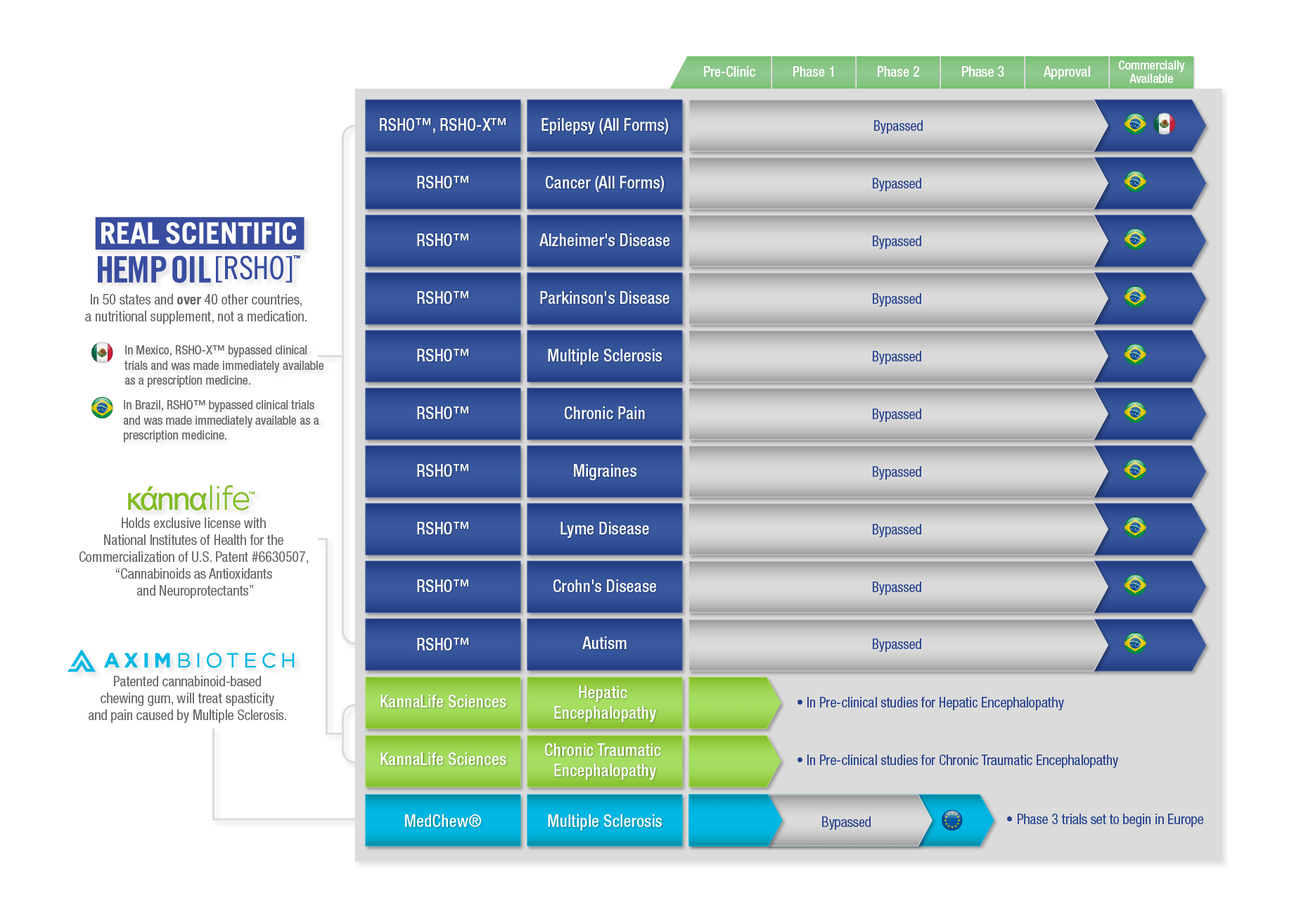
Three of our companies, AXIM Biotechnologies, Kannalife™ Sciences, and HempMeds®, makers of Real Scientific Hemp Oil™, are currently developing cannabinoid based pharmaceuticals or have hemp based medications on the market today, including the first ever legal cannabis products in Brazil and Mexico.
We invest in research to improve the health of people everywhere through the creation of innovative new applications for the cannabis plant. Research into the applications of phytocannabinoids by Medical Marijuana Inc. subsidiaries, like AXIM Biotechnologies and Kannalife Sciences, has yielded valuable new avenues for marijuana and hemp based pharmaceuticals to treat difficult and even life threatening ailments.
AXIM Biotechnologies®
Axim Biotechnologies develops cannabinoid based pharmaceuticals and owns the patent for delivery of cannabinoids through chewing gum. MedChew Rx™ is a THC and CBD based chewing gum being prepared for Phase 3 clinical trials for the relief of pain and spasticity associated with Multiple Sclerosis (MS). Once approved by the European Medicines Agency (EMA), MedChew Rx™ will be available across the European Union. With more than 2 million cases of MS occurring globally, an estimated half of all MS cases are centered in Europe, making it the single largest market for MS drugs in the world (MS Facts, 2015).
Kannalife™ Sciences
Kannalife™ Sciences is the first and currently only company licensed to commercialize products under the US patent 6630507, “Cannabinoids as antioxidants and neuroprotectants”. Cannabinoids show promise in applications as neuroprotectants and in the treatment of neurodegenerative diseases, like Alzheimer’s and Parkinson’s. CBD in particular is a strong candidate for use as a neuroprotectant due to its non-psychoactive properties. Kannalife™ Sciences has synthesized a CBD-like molecule that is both 400 times less toxic and 50 times more potent than molecular CBD, making it a more viable medical application than cannabis-derived CBD. Kannalife™ Sciences is conducting preclinical research to use this new molecule to fight Hepatic Encephalopathy (HE), a brain disease brought on by damage to the liver from abuse of alcohol and prescription pills. Although HE is considered a rare disease with fewer than 200,000 yearly cases, the cost to treat it in the U.S. alone is estimated up to $7 billion per year (Wolf, 2015). KLS-13019 could also be a promising treatment option for Chronic Traumatic Encephalopathy (CTE), the degenerative brain disease that results from repeated trauma to the head, often diagnosed in athletes (Kinney et al., 2016). Currently, CTE can only be diagnosed in post-mortem examinations of the brain. Although the development of diagnostic tests to detect early onset of CTE are being developed, there is still a need for a treatment solution.
Real Scientific Hemp Oil™
Real Scientific Hemp Oil™ from HempMeds® is currently for sale in over 40 other countries and in all 50 states, regulated in the U.S. by the FDA as a nutritional supplement. The US government continues to classify cannabis as a schedule I drug with no medical value.
Contrast that with Brazil, where, as a prescription medication, RSHO™ was able to bypass clinical trials in Brazil and is a prescription medication for nine separate indications: cancer (all forms), epilepsy (all forms), autism, Alzheimer’s disease, Parkinson’s disease, Multiple Sclerosis (MS), Crohn’s disease, chronic pain and migraines, and Lyme disease. This list of approved ailments contains some of the most debilitating and life threatening diseases we face as humans, including several where traditional medications are lacking. In Brazil, Real Scientific Hemp Oil™ can be prescribed for any indication it is deemed suitable for by a doctor, and the cost of RSHO™ is fully subsidized by the Brazilian government.
As the distributor of the first legal cannabis products imported to Brazil, HempMeds® Brasil was integral in the adoption of CBD medication in one of the top ten largest economies in the world, making RSHO™ available to more than 200 million people.
RSHO-X™ is also currently approved in Mexico as a prescription for all forms of epilepsy. Like RSHO™ in Brazil, RSHO-X™ from HempMeds® Mexico was accepted as a prescription medication without clinical trials in early 2016. Medical Marijuana, Inc. worked closely with local families to make RSHO-X™ the first legal cannabis product available in Mexico, a market of 120 million people.
There are an estimated 2.8 million cases of epilepsy in the US alone with a total of 50 million cases worldwide (WHO, 2016). 1 out of every 26 people will develop some form of epilepsy in their lifetime, making it as common as breast cancer and more common than multiple sclerosis, cerebral palsy, muscular dystrophy and Parkinson’s disease combined (Epilepsy Foundation, 2013).
References:
Epilepsy. (2016, February) World Health Organization. Retrieved from http://www.who.int/mediacentre/factsheets/fs999/en/Epilepsy Statistics. (2013, October) Epilepsy Foundation. Retrieved from http://www.epilepsy.com/learn/epilepsy-statistics
Kinney, W., McDonnell, M., Zhong, H., Liu, C., Yang, L., Ling, W., … Brenneman, D. (2016, February 10) Discovery of KLS-13019, a cannabidiol-derived neuroprotective agent, with improved potency, safety, and permeability. ACS Medicinal Chemistry Letters. doi:10.1021/acsmedchemlett.6b00009
MS Facts. (2015) European multiple sclerosis platform. Retrieved from http://www.emsp.org/about-ms/
Wolf, D. (2015, December 8) Hepatic encephalopathy. Medscape. Retrieved from http://emedicine.medscape.com/article/186101-overview






