Medical Marijuana, Inc. announced this week that AXIM® Biotechnologies secured the necessary private funding needed to continue its pharmaceutical clinical trials program in the development of cannabinoid medications for multiple medical indications.
On news of AXIM® Biotechnologies obtaining its funding, the company’s stock rose nearly 1000%, putting it in a competitive position among other cannabis-based pharmaceutical companies also pursuing clinical trials.
AXIM® Biotechnologies’ recent financing permits the company to continue in the research and development of its innovative pharmaceutical delivery systems and hemp-based pharmaceutical ingredients for the treatment of intractable conditions for which there is currently no effective remedy.
MedChew® Rx Tested for Multiple Sclerosis
MedChew® Rx, a cannabinoid-infused chewing gum aimed at treating the pain and spasticity in multiple sclerosis (MS), will soon undergo clinical trials in Europe.
AXIM® Biotechnologies will be conducting their clinical trial on MedChew® Rx at the Free University of Amsterdam, The Netherlands. The study will be performed in collaboration with the University of Plymouth, UK, and an academic center in the U.S. The anticipated duration of the clinical trials prior to FDA/ EMA registration is approximately twelve months.
“We’re extremely pleased that AXIM has received the financing it needs to continue clinical efforts with the Company’s two near-term prospects. Further, the financing will allow the AXIM team of visionaries and world leaders, with proven track records, to create further cannabinoid-based innovations that may address additional indications,” said Medical Marijuana, Inc. Chief Executive Officer Dr. Stuart Titus.
AXIM® Biotechnologies is looking to secure MedChew® Rx as a novel, patented delivery method for cannabinoids in the treatment of chronic pain and spasticity for patients with multiple sclerosis. The potential economic impact for MedChew® Rx is estimated at $4.8 billion USD for these symptoms of MS.
CanChew® Plus May Treat Irritable Bowel Syndrome
The company has also begun studies involving its cannabinoid-infused CanChew® Plus Gum. The goal of these studies is to begin to determine the potential reduction of irritable bowel syndrome (IBS) symptoms in suffering patients.
“We congratulate the AXIM team on reaching another major milestone in the Company’s continued efforts to research new solutions using cannabinoid-based drugs,” said Dr. Stuart Titus, CEO of Medical Marijuana, Inc. “We are very excited about the new formulation of CanChew Plus, which we feel will offer superior bioavailability versus our prior rendition of CanChew.”
Wageningen University in the Netherlands is slated to conduct the CanChew® Plus studies. The study will test 10 mg and 30 mg single dose applications for the concentration of CBD in the blood after 30 minutes. This early step will determine the administered patient dose for AXIM® Biotechnologies’ Phase II trials to follow.
Topically Applied CBG Ointment for Psoriasis and Eczema
AXIM® Biotechnologies is also undertaking human dermatological clinical trials based on the company’s proprietary, patent-pending topical ointment formulation AX-1602, which includes the “stem cell cannabinoid” cannabigerol (CBG) and other cannabinoids. The topical application of CBG is being tested for efficacy for the skin conditions psoriasis and atopic dermatitis (eczema).
AXIM’s CEO, Dr. George Anastassov, states, “We are extremely pleased that our ongoing efforts in finding solutions to conditions for which there are no effective treatments… are culminating with the clinical trials of our Renecann™ product. Psoriasis and atopic dermatitis are debilitating conditions causing significant decrease in quality of life. Utilizing our proprietary technologies and incorporating unique cannabinoids, we believe that we will be able to effectively address these concerns.”
The results of the AX-1602 study will determine whether the company pursues further clinical trials and a new drug application or moves to make the product available over-the-counter.
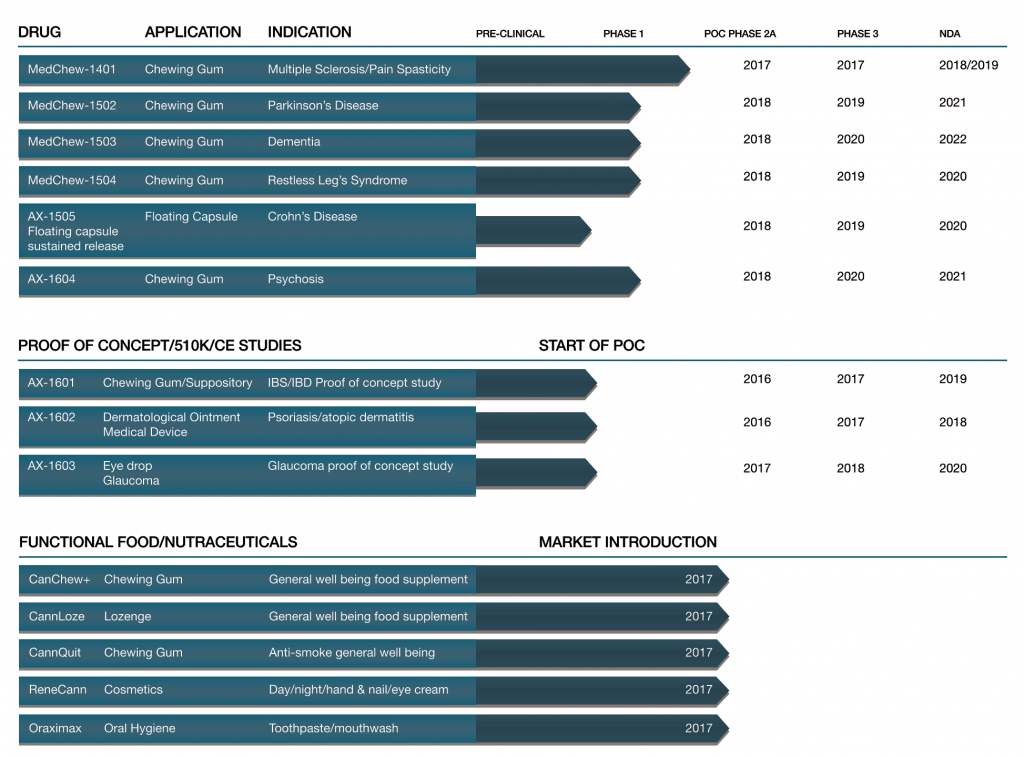
AXIM Releases New Drug Timelines
AXIM® Biotechnologies recently released a drug pipeline chart revealing their anticipated clinical trials. The infographic highlights the timelines established by the company for clinical trials and new drug applications.
“We commend AXIM for publishing a product pipeline chart on its website as it provides potential patients and investors with critical Company information about the numerous cannabinoid-based therapeutic products that AXIM currently has undergoing clinical trials,” said Medical Marijuana, Inc. Chief Executive Officer Dr. Stuart Titus. “It’s exciting to see the actual timelines for these cannabinoid-based innovations currently in development, as we’re confident that once on the market these novel therapeutics will provide consumers with a wide variety of new options worldwide.”
This clearly developed graphic shows what stage of development each AXIM® Biotechnologies product is in and gives schedules and estimated dates for completion and market introduction. As the medical marijuana industry matures, pharmaceutical applications of cannabinoids will continue to advance as well. AXIM® Biotechnologies’ aggressive pursuit of clinical trials will secure their vigorous position in the cannabinoid-based pharmaceutical market.